
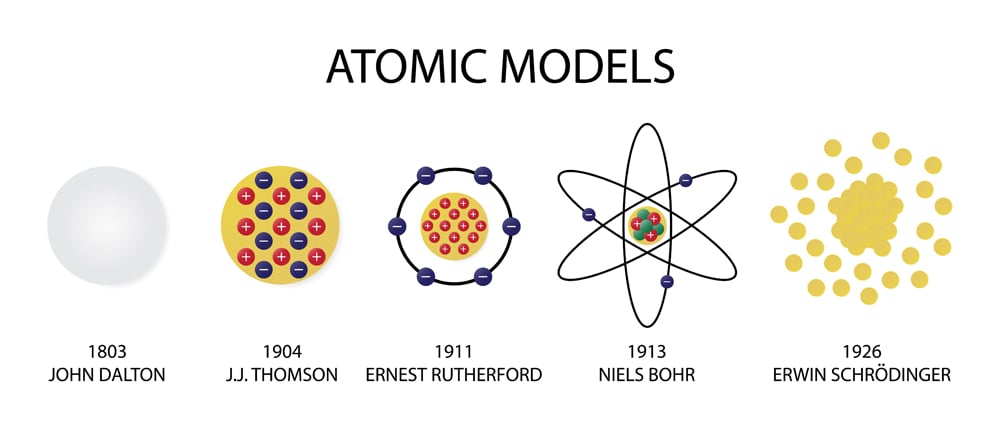
The model did not account for the fact that electrons have a wave-like nature, which was later revealed by quantum mechanics.The model could not explain the existence of isotopes, which are atoms of the same element that have a different number of neutrons in their nuclei.It could not explain the phenomenon of radioactivity.It could not explain the observed phenomena of atomic spectra.
It could not explain the stability of atoms, as the negatively charged electrons should have been attracted to the positively charged soup and collapsed into the center.

Thomson’s model was able to explain some previously unexplained phenomena, such as the fact that atoms are electrically neutral, and that the ratio of the charge of an electron to its mass is the same for all elements.ġ.2.1 Limitations of J.J Thomson’s Model: He proposed that atoms were neutral because the negative charges of the electrons were balanced out by an equal amount of positive charge in the soup. He had discovered the electron, a subatomic particle while conducting experiments on cathode rays. In this model, Thomson proposed that atoms were composed of negatively charged electrons embedded in a positively charged “soup” or “pudding”.
Thomson’s atomic model, also known as the “plum pudding” model, was proposed in 1904. All of these limitations and errors in Dalton’s atomic model were later addressed by more advanced theories, such as quantum mechanics and the electron cloud model, which provided a more detailed and accurate understanding of atomic structure and behavior.Dalton’s atomic theory also did not have any concept of chemical bonding, which is the way atoms combine to form molecules.The model assumed that atoms were indestructible, which was later disproved by the discovery of nuclear reactions, which can break atoms apart or merge them together to create new elements.The model did not account for the existence of subatomic particles- electrons, protons, and neutrons, which were later discovered by scientists such as J.J.The model did not explain the phenomenon of atomic spectra, which was observed in light emitted or absorbed by atoms.The model did not account for the existence of isotopes- that are atoms of the same element but they have a different number of neutrons in their nuclei.It did not explain the internal structure of atoms, which was later revealed by more advanced theories such as the atomic models of Niels Bohr and Erwin Schrödinger.There are several demerits or limitations of Dalton’s atomic model: However, Dalton’s model did not explain the internal structure of atoms, and it was later replaced by more advanced theories that provided a more detailed understanding of atomic structure and behavior. They are in constant motion and collide with one another, causing changes in their motion and resulting in chemical reactions. In this model, atoms are like billiard balls, which are thought to be solid and indestructible. Thomson’s Atomic Model or The Plum Pudding Modelĭalton’s Billiard Ball Model, proposed by John Dalton in 1803, is an early atomic theory that describes the behavior of matter as being composed of small, indivisible particles called atoms.Although the existence of atoms has long been recognized. Many different atomic models have existed throughout the history of atomic physics.
